
Hello! I am Tawsif. My research focuses on the detection, monitoring and remediation of pathogens and PFAS in food and water. Beside research, I love anime, music and traveling.

Hello! I am Tawsif. My research focuses on the detection, monitoring and remediation of pathogens and PFAS in food and water. Beside research, I love anime, music and traveling.

Position: Graduate Research Assistant
E-mail: u1146520@utah.edu
Lab: 3800 SMBB
Lab Name: State of Utah Center of Excellence for Biomedical Microfluidics
Supervisor: Dr. Bruce Gale
Education
Ph.D. Electrical and Computer Engineering, University of Utah, Salt Lake City, Utah, 2023 (ongoing)
M.Sc. Information and Communication Technology, Bangladesh University of Engineering and Technology, Dhaka, Bangladesh, 2017
B.Sc. Electrical, Electronics and Communication Engineering, Military Institute of Science and Technology, Dhaka, Bangladesh, 2011
Research Projects :
1. Automated passive serial dilution microfluidic chip for calcium quantification based on the Arsenazo III method :
In this study, a valveless, resistance-based method was used to design and develop a passive microfluidic chip to implement an automated calcium assay in urine samples using the arsenazo III method with a 3D printed mold. The resistive design of the channels provides a precise mixing in the assay at a ratio of 250:25:2 for water, reagents, and samples, respectively, with an added advantage of reduced process cost due to the 3D printed mold. The reaction between arsenazo III and calcium in the urine results in a purple-colored complex that is quantified using a visible light source spectrophotometer, a portable fiber-optic spectrophotometer with a UV (ultraviolet) source and a smartphone camera capable of capturing RGB values. A linear range from 1.62-9.59 mg/dL (Limit of Detection: 0.205 mg/dL) for calcium concentration in urine was obtained using a visible light spectrophotometer with R2=0.99. Similarly, the linear range obtained for the portable spectrometer and smartphone camera was 1.62-6.50 mg/dL with R2=0.93 and R2=0.97, respectively. The chip fabrication method and assay were found to be highly reproducible (σ<0.001). The approach becomes highly portable with the use of a smartphone camera for quantification. The proposed microfluidic system can successfully quantify calcium in a relevant range in urine samples and is sensitive enough for an automated urinalysis system inside a smart toilet. Additionally, it should also be applicable to other assays by changing channel resistance according to the desired dilution or mixing ratio.
Publication :
N. Tazin, D. Patel, C. J. Lambert, M. H. M. Shad, J. Campbell, and B. K. Gale, “Automated passive serial dilution microfluidic chip for calcium quantification based on the Arsenazo III method,” Sensors & Diagnostics, vol. 1, no. 4, pp. 810–820, 2022, doi: 10.1039/d2sd00022a
2. Transgenic expression in zebrafish embryos with an intact chorion by electroporation and microinjection :
Electroporation is regularly used to deliver agents into cells, including transgenic materials, but it is not used for mutating zebrafish embryos due to the lack of suitable systems, information on appropriate operating parameters, and the challenges posed by the protective chorion. Here, a novel method for gene delivery in zebrafish embryos was developed by combining microinjection into the space between the chorion and the embryo followed by electroporation. This method eliminates the need for chorion removal and injecting into the space between the chorion and embryo eliminates the need for finding and identifying key cell locations before performing an injection, making the process much simpler and more automatable. We also developed a microfluidic electroporation system and optimized electric pulse parameters for transgenesis of embryos. The study provided a novel method for gene delivery in zebrafish embryos that can be potentially implemented in a high throughput transgenesis or mutagenesis system.
Publication :
N. Tazin, C. J. Lambert, R. Samuel, T. J. Stevenson, J. Bonkowsky and B. K. Gale, “Transgenic expression in zebrafish embryos with an intact chorion by electroporation and microinjection,” (Submitted to Biotechnology Reports)
3. Optimization of an automated system (ZEG) for rapid cellular extraction from live zebrafish :
Conducting large-scale screening of mutagenesis or identifying transgenic fish can be a challenging and time-consuming process for researchers due to the absence of automated genotyping tools for live animals. Zebrafish present significant challenges to researchers due to the lack of automated genotyping tools, forcing them to rely on manual labor-intensive methods. This limitation restricts potential drug screening options, reduces the ability to analyze embryonic and larval phenotypes, and necessitates breeding additional animals to ensure the desired genotype. To overcome this limitation, researchers are exploring the development of automated genotyping tools for live animals that would significantly improve the efficiency and accuracy of genetic screening in zebrafish and other species. The focus of this research was to optimize the Zebrafish Embryo Genotyper (ZEG), an automated system utilized for the rapid extraction of cellular material from zebrafish embryos. The aim was to improve the efficiency of DNA collection from the chip by identifying the key factors that contribute to the process. To resolve the problem of sample solution evaporation from the chip wells, the chip design underwent optimization. This enhancement effectively eliminated the issue of evaporation and had minimal impact on the system’s overall performance.
4. An electroporation system to improve the fish embryo toxicity test with zebrafish :
The zebrafish model has emerged as a useful model for biomedical research and extended its application for environmental toxicology studies. However, the presence of chorion during the embryo stage limits the exposure to toxic elements and creates the possibility of a false-negative outcome in Fish Embryo Toxicity Testing (FET). This paper presents electroporation as a technique to improve the delivery of toxic elements inside the chorion for exposing the fish embryos to the material at an early stage. A custom-made electroporation device has been developed to place embryos and electrodes with required electric circuitry that provides suitable pulses for creating a sufficient electric field for aiding the entry of molecules inside the chorion. The optimized parameters facilitate material inside the chorion without affecting the survival rate of the embryos. The effectiveness of the proposed system with electroporation as a technique has been demonstrated using trypan blue dye and gold nanoparticles (Au-NP, 20-40 nm). The result demonstrated the feasibility of controlling the concentration of dye and nanoparticles delivered inside the chorion by optimizing the pulse parameters like pulse width, pulse number, and amplitude. Silver nanoparticles (Ag-NP, 10 nm) have been used as a toxic element to determine phenotypical changes due to electroporation aiding the material delivery inside the chorion. The toxicity effect of mortality, heart rate, and phenotypic defects was recorded.. This electroporation approach in Fish Embryo Toxicity Testing can provide potential usage in toxicity testing and drug delivery.
5. A Simulation study of the electrical model Electroporation for Zebrafish Embryos :
In this paper, we have performed electroporation with zebrafish embryos at the age of 0 hours of post fertilization with and without removing the chorion. Before applying voltage pulses, we injected the genetic material inside the chorion. To understand its limitations, we have modelled electroporation for a zebrafish embryo by using COMSOL Multiphysics software and determined.

 I received my Bachelor’s degree from K.N. Toosi University of Technology and Master’s degree from Sharif University of Technology both in Mechanical Engineering. Currently, I am pursuing a PhD in Mechanical Engineering at the University of Utah.
I received my Bachelor’s degree from K.N. Toosi University of Technology and Master’s degree from Sharif University of Technology both in Mechanical Engineering. Currently, I am pursuing a PhD in Mechanical Engineering at the University of Utah.
My work focuses on two projects: 1. separation of virus like particles; 2. Characterization, purification and sorting of exosomes and oncosomes.
Separation of virus like particles: this research focuses on the separation and characterization of different types of recombinant protein-based virus like particles (VLPs). In addition, the separation of the monomers from aggregates using electric field flow fractionation (ElFFF) and asymmetric flow field flow fractionation (AF4). VLPs play an important role medicine. They are popularly researched as potential drug carriers, and safe vaccines.
Characterization, purification, and sorting of exosomes and oncosomes: this project is concerning about the characterization, isolation, purification, and sorting of exosomes and oncosomes derived from cell culture supernatants and plasma samples.
Interests:
On paper, I’m probably your prototypical “nerd”. I’ve been parts of clubs like the Jedi knights club (Yes, we actually had choreographed Jedi light saber battles. yes, be jealous.), LARPing (Live action role play) club, watch anime, and have participated in several zombie apocalypse exercises. Judge me as you see fit.
Research Project: Pathogen detection project. My project aims to develop an ultra sensitive device that is capable of detecting pathogens in water, food, and other media. Our device must meet several specs: it must be ultra sensitive (detecting well below the limits of infectivity for target pathogens), specific (only detect target pathogens), Multiplexible (detects a wide array of predetermined pathogen targets in one medium ), and finally rapid (able to do all this in roughly 2 hours).
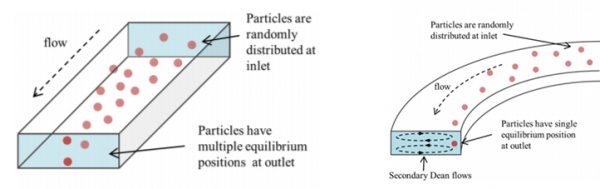
I received My Master in theoretical condensed matter physics and later shifted my attention to experimental and computational biophysics. While I maintain an interest in these subjects, more recently I have become fascinated by the possibility of using microfluidics approaches in biology, medicine and beyond. For the purpose of improving health care quality and designing applicable public health interventions, I joined a microfluidics group in the Department of Mechanical Engineering run by Dr. Bruce Gale as a Ph.D. student. My thesis work centers around trying to investigate the separation dynamics of bio-particles. I focus particularly on sperm, which has shown unusual behavior in the spiral channel using particle simulations. This will improve the understanding of the underlying mechanisms of particle sorting. This information can be leveraged to design sorting devices for effective separation of other asymmetrical and non-spherical bio-particles in the spiral channel device.
My research focuses on two distinct projects:
I just started my 2nd year of my biomedical engineering Ph.D. and am currently working on 2 projects for Dr. Gale in collaboration with Dr. Jay Agarwal. The vascular coupler device is an alternative to hand sutures for arterial anastomosis. The coupler utilizes a backpack type clip to allow surgeons to quickly connect two ends of a severed artery or vein back together. I’m also working on a nerve conduit device for use in peripheral nerve repair and regeneration. The conduit provides structure as well as a system for drug delivery during axon regeneration after injury.
Research: Vascular Coupling Device and Nerve Conduit device
I am interested in microfluidic cell culture devices known as organs-on-chips, which are used to model physiological functions of tissues and organs. These devices have the potential to better mimic human physiology and disease compared with traditional cell cultures or animal-based assays, and therefore have significant promise for future drug development. Outside of work, I’m interested in hiking, playing piano and guitar, cooking, and gaming.
University of Utah Mechanical Engineering Ph.D. student, Michael Beeman, advised by Mechanical Engineering professor Bruce Gale, is an alumni of the colorado school of mines where he received a masters of science in Mechanical Engineering. My work focuses on two distinct projects: pathogen detection using electrochemistry and glass nano-pores.
Bacteria Detection: This project focuses on the use of electrochemical signal processing to detect pathogens in liquid and solid food sources. The project is done in collaboration with the United States Army and with the Salt Lake-based company Espira.
Glass Nano-pore Reduction: I also work on a project researching deposition techniques to reduce the pore diameter of glass nano-pores. The project is in conjunction with Electronic Bioscience Inc.

I work with the Microfluidic Flow Cell Array (MFCA). The MFCA is a High troughput microfluidic system that allows for the parallel testing of different compounds via continuous flow. This system can be used to test 2D (single cell cultures) and 3D (frozen/alive cryo sectioned tissue slices, organoids) models that are more physiologically accurate than existing models. This system aims to improve the “weeding out” of candidates in the early stages of the drug development process. Thus, saving time and expensive resources.
I love playing every sport out there. EXCEPT soccer. I love nature though I’m not a big fan of camping. Reading a book by the ocean is the definition of a perfect day for me.